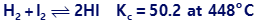Chapter 13: Q87E (page 760)
Question: What is the minimum mass of CaCO3 required to establish equilibrium at a certain temperature in a 6.50-L container if the equilibrium constant (Kc) is 0.050 for the decomposition reaction of CaCO3 at that temperature?
Short Answer
The minimum mass of CaCO3required is 3.25 g.