Chapter 13: Q85E (page 760)
Question: Calculate the number of grams of HI that are at equilibrium with 1.25 mol of H2 and 63.5 g of iodine at 448°C.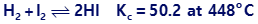
Short Answer
The mass of HI is 507 g.
Learning Materials
Features
Discover
Chapter 13: Q85E (page 760)
Question: Calculate the number of grams of HI that are at equilibrium with 1.25 mol of H2 and 63.5 g of iodine at 448°C.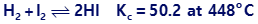
The mass of HI is 507 g.
All the tools & learning materials you need for study success - in one app.
Get started for free
Write the expression of the reaction quotient for the ionization of HOCN in water.
Explain how to recognize the conditions under which changes in pressure would affect systems at equilibrium.
How can the pressure of water vapor are increased in the following equilibrium?
\({H_2}O(l) \rightleftharpoons {H_2}O(g)\) \(\Delta H = 41kJ\)
Question: Calculate the pressures of NO, Cl2, and NOCl in an equilibrium mixture produced by the reaction of a starting mixture with 4.0 atm NO and 2.0 atm Cl2. (Hint: KP is small; assume the reverse reaction goes to completion then comes back to equilibrium.)
A sample of ammonium chloride was heated in a closed container. NH4 Cl (s)⇌ NH3 (g) + HCl(g)at equilibrium, the pressure of NH3 (g)was found to be 1.75 atm. What is the value of the equilibrium constant, Kp, for the decomposition at this temperature?
What do you think about this solution?
We value your feedback to improve our textbook solutions.