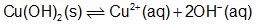Chapter 15: Q64E (page 876)
Question: Explain why addition of \(N{H_3}\;or\;HN{O_3}\) to a saturated solution of \(Cu{(OH)_2}\)in contact with solid \(Cu{(OH)_2}\) increases the solubility of the solid.
Short Answer
When \({\rm{N}}{{\rm{H}}_3}\) react with \({\rm{C}}{{\rm{u}}^{2 + }}\)This will decrease the concentration of \({\rm{C}}{{\rm{u}}^{2 + }}\) ions, and move equilibrium to the right. The concentration of \({\rm{O}}{{\rm{H}}^ - }\). will increase, and the amount of the solid \({\rm{Cu}}{({\rm{OH}})_2}\) will decrease. Hence, the solubility of \({\rm{Cu}}{({\rm{OH}})_2}\) is increased.
When \({\rm{HN}}{{\rm{O}}_3}\)dissociates into \({{\rm{H}}^ + }{\rm{and\;N}}{{\rm{O}}_3} - \) ions. \({{\rm{H}}^ + }\)ions react with \({\rm{O}}{{\rm{H}}^ - }\) ion, to produce \({{\rm{H}}_2}{\rm{O}}.\)This will decrease the concentration of \({\rm{O}}{{\rm{H}}^ - }\)ions, and move equilibrium to the right. The concentration of \({\rm{C}}{{\rm{u}}^{2 + }}\)will increase, and the amount of the solid \({\rm{Cu}}{({\rm{OH}})_2}\)will decrease. Hence, the solubility of \({\rm{Cu}}{({\rm{OH}})_2}\)is increased.