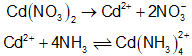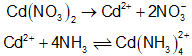
Given,
\(1 - 0.100{\rm{L\;of\;}}0.0100{\rm{MCd}}{\left( {{\rm{N}}{{\rm{O}}_3}} \right)_2}\)
And \(1.150{\rm{L\;of\;}}0.100{\rm{MN}}{{\rm{H}}_3}\)
\({K_f}{\rm{\;for\;Cd}}\left( {{\rm{N}}{{\rm{H}}_3}} \right)_4^{2 + }{\rm{\;is\;}}1.3 \cdot {10^7}\)
Calculate number of moles of \({\rm{Cd}}{\left( {{\rm{N}}{{\rm{O}}_3}} \right)_2}{\rm{\;and\;N}}{{\rm{H}}_3}\),
\(\begin{array}{*{20}{c}}{{n_{{\rm{Cd}}{{\left( {N{O_3}} \right)}_2}}} = 0.100{\rm{L}} \cdot 0.0100{\rm{M}}}\\{ = 0.001{\rm{mol}}}\end{array}\)
\({n_{{\rm{N}}{{\rm{H}}_3}}} = 1.15{\rm{L}} \cdot 0.100{\rm{M}} = {\bf{0}}.{\bf{115}}{\rm{mol}}\)
\(0.001{\rm{\;mol of\;Cd}}{\left( {{\rm{N}}{{\rm{O}}_3}} \right)_2}\)dissociates into \(0.001{\rm{molC}}{{\rm{d}}^{2 + }}\) and \(0.002{\rm{molNO}}_3^ - \)
\(0.001{\rm{mol\;of\;C}}{{\rm{d}}^{2 + }}\)will react with \(0.004{\rm{mol\;of\;N}}{{\rm{H}}_3}\)and produce \({\bf{0}}.{\bf{001}}{\rm{\;mol of\;Cd}}{\left( {{\rm{N}}{{\rm{H}}_3}} \right)_4}\)
So, excess of \({\rm{N}}{{\rm{H}}_3}\)is \(0.115{\rm{mol}} - 0.004{\rm{mol}} = {\bf{0}}.{\bf{111}}{\rm{mol}}\).