Chapter 3: Q.25E (page 129)
Determine which of the following contains the greatest mass of aluminum: 122 g of AlPO4, 266 g of Al2C16, or 225 g of Al2S3.
Short Answer
Greatest mass of Al is in Al2S3 = 81 g.
Learning Materials
Features
Discover
Chapter 3: Q.25E (page 129)
Determine which of the following contains the greatest mass of aluminum: 122 g of AlPO4, 266 g of Al2C16, or 225 g of Al2S3.
Greatest mass of Al is in Al2S3 = 81 g.
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine the number of moles of compound and the number of moles of each type of atom in each of the following:
(a) 25.0 g of propylene, C3H6
(b) 3.06×10−3 g of the amino acid glycine, C2H5NO2
(c) 25 lb of the herbicide Treflan, C13H16N2O4F (1 lb = 454 g)
(d) 0.125 kg of the insecticide Paris Green, Cu4(AsO3)2(CH3CO2)2
(e) 325 mg of aspirin, C6H4(CO2H) (CO2CH3)
Question: If 4.12L of a 0.850M-H3PO4 solution is be diluted to a volume of 10.00L, what is the concentration of the resulting solution?
Determine the molecular mass of the following compounds:
(a)
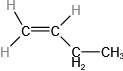
(b)
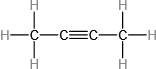
(c)
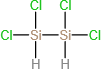
(d)
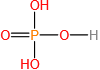
47. Determine the molarity for each of the following solutions:
(a) 0.444 mol of CoCl2 in 0.654 L of solution
(b) 98.0 g of phosphoric acid, H3PO4, in 1.00 L of solution
(c) 0.2074 g of calcium hydroxide, Ca(OH)2, in 40.00 mL of solution
(d) 10.5 kg of Na2SO4∙10H2O in 18.60 L of solution
(e) 7.0×10-3 mol of I2 in 100.0 mL of solution
(f) 1.8 × 104 mg of HCl in 0.075 L of solution
Question: A tube of toothpaste contains 0.76 g of sodium monofluorophosphate (Na2PO3F) in 100 mL.
(a) What mass of fluorine atoms in mg was present?
(b) How many fluorine atoms were present?
What do you think about this solution?
We value your feedback to improve our textbook solutions.