Chapter 7: Q65E (page 400)
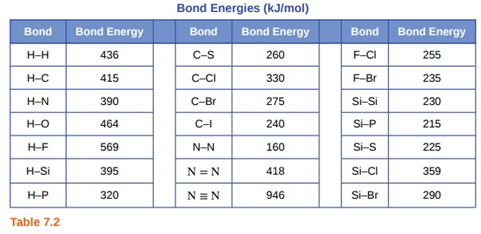
Question: Using the bond energies in Table \({\rm{7}}{\rm{.2}}\), determine the approximate enthalpy change for each of the following reactions:
(a) \({{\rm{H}}_{\rm{2}}}{\rm{(g) + B}}{{\rm{r}}_{\rm{2}}}{\rm{(g)}} \to {\rm{2HBr(g)}}\)
(b) \({\rm{C}}{{\rm{H}}_{\rm{4}}}{\rm{(g) + }}{{\rm{I}}_{\rm{2}}}{\rm{(g)}} \to {\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{I(g) + HI(g)}}\)
(c) \({{\rm{C}}_{\rm{2}}}{{\rm{H}}_4}{\rm{(g) + 3}}{{\rm{O}}_{\rm{2}}}{\rm{(g)}} \to {\rm{2C}}{{\rm{O}}_{\rm{2}}}{\rm{(g) + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O(g)}}\)
Short Answer
(a) For the reaction \({{\rm{H}}_{\rm{2}}}{\rm{(g) + B}}{{\rm{r}}_{\rm{2}}}{\rm{(g)}} \to {\rm{2HBr(g)}}\), the value for enthalpy change is \({\rm{\Delta H}}_{{\rm{298}}}^{\rm{^\circ }}{\rm{ = - 144\;kJ}}\).
(b) For the reaction\({\rm{C}}{{\rm{H}}_{\rm{4}}}{\rm{(g) + }}{{\rm{I}}_{\rm{2}}}{\rm{(g)}} \to {\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{I(g) + HI(g)}}\), the value for enthalpy change is\({\rm{\Delta H}}_{{\rm{298}}}^{\rm{^\circ }}{\rm{ = 30\;kJ}}\).
(c) For the reaction \({{\rm{C}}_{\rm{2}}}{{\rm{H}}_4}{\rm{(g) + 3}}{{\rm{O}}_{\rm{2}}}{\rm{(g)}} \to {\rm{2C}}{{\rm{O}}_{\rm{2}}}{\rm{(g) + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O(g)}}\), the value for enthalpy change is \({\rm{\Delta H}}_{{\rm{298}}}^{\rm{^\circ }}{\rm{ = - 1055\;kJ}}\).

