Chapter 7: Q103E (page 405)
Is the \({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{BBC}}{{\rm{l}}_{\rm{2}}}\)molecule polar or nonpolar?
Short Answer
The molecule \({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{BBC}}{{\rm{l}}_{\rm{2}}}\) is nonpolar.
Learning Materials
Features
Discover
Chapter 7: Q103E (page 405)
Is the \({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{BBC}}{{\rm{l}}_{\rm{2}}}\)molecule polar or nonpolar?
The molecule \({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{BBC}}{{\rm{l}}_{\rm{2}}}\) is nonpolar.
All the tools & learning materials you need for study success - in one app.
Get started for free
There are three possible structures for \({\rm{PC}}{{\rm{l}}_{\rm{2}}}{\rm{\;}}{{\rm{F}}_{\rm{3}}}\)with phosphorus as the central atom. Draw them and discuss how measurements of dipole moments could help distinguish among them.
Question: Using the standard enthalpy of formation data in Appendix G, show how the standard enthalpy of formation of \({\rm{HCl(g)}}\) can be used to determine the bond energy.
From their positions in the periodic table, arrange the atoms in each of the following series in order of increasing electronegativity: (a)\({\rm{C, F, H, N, O}}\)(b)\({\rm{Br, Cl, F, H, I }}\)(c)\({\rm{F, H, O, P, S }}\)(d)\({\rm{Al, H, Na, O, P}}\)(e)\({\rm{Ba, H, N, O, As}}\).
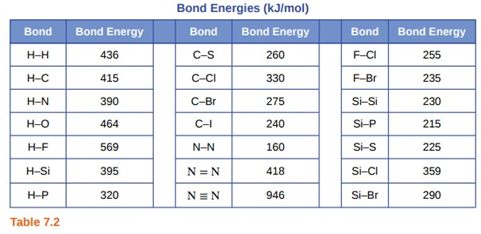
Question: Using the bond energies in Table \({\rm{7}}{\rm{.2}}\), determine the approximate enthalpy change for each of the following reactions:
(a) \({{\rm{H}}_{\rm{2}}}{\rm{(g) + B}}{{\rm{r}}_{\rm{2}}}{\rm{(g)}} \to {\rm{2HBr(g)}}\)
(b) \({\rm{C}}{{\rm{H}}_{\rm{4}}}{\rm{(g) + }}{{\rm{I}}_{\rm{2}}}{\rm{(g)}} \to {\rm{C}}{{\rm{H}}_{\rm{3}}}{\rm{I(g) + HI(g)}}\)
(c) \({{\rm{C}}_{\rm{2}}}{{\rm{H}}_4}{\rm{(g) + 3}}{{\rm{O}}_{\rm{2}}}{\rm{(g)}} \to {\rm{2C}}{{\rm{O}}_{\rm{2}}}{\rm{(g) + 2}}{{\rm{H}}_{\rm{2}}}{\rm{O(g)}}\)
How are single, double, and triple bonds similar? How do they differ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.