Chapter 7: Chemical Bonding and Molecular Geometry
Q24 E
Many monatomic ions are found in seawater, including the ions formed from the following list of elements. Write the Lewis symbols for the monatomic ions formed from the following elements: (a)\({\rm{CI}}\)(b)\({\rm{Na}}\)(c)\({\rm{Mg}}\)(d)\({\rm{Ca}}\)(e)\({\rm{K}}\)(f)\({\rm{Br}}\)(g)\({\rm{Sr}}\)(h)\({\rm{F}}\).
Q25 E
Write the Lewis symbols of the ions in each of the following ionic compounds and the Lewis symbols of the atom from which they are formed: (a) \({\rm{MgS}}\) (b) \({\rm{A}}{{\rm{l}}_{\rm{2}}}{{\rm{O}}_{\rm{3}}}\) (c) \({\rm{GaC}}{{\rm{l}}_{\rm{3}}}\) (d) \({{\rm{K}}_{\rm{2}}}{\rm{O}}\) (e) \({\rm{L}}{{\rm{i}}_{\rm{3}}}{\rm{N}}\) (f) \({\rm{KF}}\) .
Q26 E
In the Lewis structures listed here, M and X represent various elements in the third period of the periodic table. Write the formula of each compound using the chemical symbols of each element:
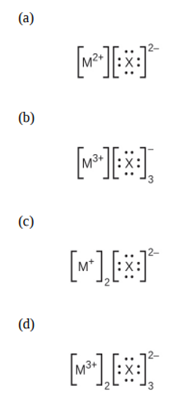
Q27 E
Write the Lewis structure for the diatomic molecule \({{\rm{P}}_{\rm{2}}}\), an unstable form of phosphorus found in high temperature phosphorus vapor.
Q29 E
Write Lewis structures for the following: (a)\({{\rm{O}}_{\rm{2}}}\)(b)\({{\rm{H}}_{\rm{2}}}{\rm{CO}}\)(c)\({\rm{As}}{{\rm{F}}_{\rm{3}}}\)(d)\({\rm{ClNO}}\)(e)\({\rm{SiC}}{{\rm{l}}_{\rm{4}}}\)(f)\({{\rm{H}}_{\rm{3}}}{{\rm{O}}^{\rm{ + }}}\)(g)\({\rm{N}}{{\rm{H}}_{\rm{4}}}^{\rm{ + }}\)(h)\({\rm{B}}{{\rm{F}}_{\rm{4}}}^{\rm{ - }}\)(i)\({\rm{HCCH}}\)(j)\({\rm{CICN}}\)(k)\({{\rm{C}}_{\rm{2}}}^{{\rm{2 + }}}\).
Q2E
Iron(\({\rm{III}}\)) sulfate (\({\rm{F}}{{\rm{e}}_{\rm{2}}}{{\rm{(S}}{{\rm{O}}_{\rm{4}}}{\rm{)}}_{\rm{3}}}\)) is composed of \({\rm{F}}{{\rm{e}}^{{\rm{3 + }}}}\) and \({\rm{S}}{{\rm{O}}_{\rm{4}}}^{{\rm{2 - }}}\) ions. Explain why a sample of iron(\({\rm{III}}\)) sulfate is uncharged.
Q30 E
Write Lewis structures for the following: (a) \({\rm{CI}}{{\rm{F}}_{\rm{3}}}\) (b) \({\rm{PC}}{{\rm{I}}_{\rm{5}}}\) (c) \({\rm{B}}{{\rm{F}}_{\rm{3}}}\) (d) \({\rm{P}}{{\rm{F}}_{\rm{6}}}^{\rm{ - }}\) .
Q31 E
Write Lewis structures for the following: (a)\({\rm{Se}}{{\rm{F}}_{\rm{6}}}\)(b)\({\rm{Xe}}{{\rm{F}}_{\rm{4}}}\)(c)\({\rm{SeC}}{{\rm{l}}_{\rm{3}}}^{\rm{ + }}\)(d)\({\rm{C}}{{\rm{l}}_{\rm{2}}}{\rm{BBC}}{{\rm{l}}_{\rm{2}}}\)(contains a\({\rm{B - B}}\)bond).
Q32 E
Write Lewis structures for: (a) \({\rm{P}}{{\rm{O}}_{\rm{4}}}^{{\rm{3 - }}}\) (b) \({\rm{IC}}{{\rm{I}}_{\rm{4}}}^{\rm{ - }}\) (c) \({\rm{S}}{{\rm{O}}_{\rm{3}}}^{{\rm{2 - }}}\) (d) \({\rm{HONO}}\) .
Q33 E
Correct the following statement: “The bonds in solid\({\text{PbC}}{{\text{I}}_{\text{2}}}\)are ionic; the bond in a\({\text{HCl}}\)molecule is covalent. Thus, all of the valence electrons in\({\text{PbC}}{{\text{I}}_{\text{2}}}\)are located on the\({\text{C}}{{\text{I}}^{\text{ - }}}\)ions, and all of the valence electrons in a\({\text{HCl}}\)molecule are shared between the\({\text{H}}\)and\({\text{CI}}\)atoms.”
