Chapter 7: Chemical Bonding and Molecular Geometry
Question 45 E
(a) sulphur dioxide, \({\rm{S}}{{\rm{O}}_{\rm{2}}}\)
(b) carbonate ion, \({\rm{CO}}_{\rm{3}}^{{\rm{2 - }}}\)
(c) hydrogen carbonate ion, \({\rm{HCO}}_{\rm{3}}^{\rm{ - }}\) (\({\rm{C}}\) is bonded to an \({\rm{OH}}\) group and two \({\rm{O}}\) atoms)
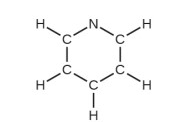
(d) pyridine:
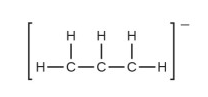
(e) the allyl ion:
Question 46 E
Write the resonance forms of ozone, \({{\rm{O}}_{\rm{3}}}\), the component of the upper atmosphere that protects the Earth from ultraviolet radiation.
Question 48 E
In terms of the bonds present, explain why acetic acid, \(C{H_3}C{O_2}H\), contains two distinct types of carbon-oxygen bonds, whereas the acetate ion, formed by loss of a hydrogen ion from acetic acid, only contains one type of carbon-oxygen bond. The skeleton structures of these species are shown:
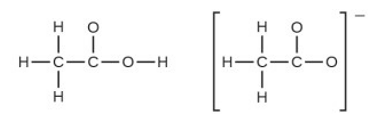
Question 49 E
Write the Lewis structures for the following, and include resonance structures where appropriate. Indicate which has the strongest carbon-oxygen bond.
(a) \(C{O_2}\)
(b) \(CO\)
Question 51 E
Determine the formal charge of each element in the following:
(a) \({\rm{HCl}}\)
(b) \({\rm{C}}{{\rm{F}}_{\rm{4}}}\)
(c) \({\rm{PC}}{{\rm{l}}_{\rm{3}}}\)
(d) \({\rm{P}}{{\rm{F}}_{\rm{5}}}\)
Question 52 E
Determine the formal charge of each element in the following:
(a) \({{\rm{H}}_{\rm{3}}}{{\rm{O}}^{\rm{ + }}}\)
(b) \({\rm{SO}}_{\rm{4}}^{{\rm{2 - }}}\)
(c) \({\rm{N}}{{\rm{H}}_{\rm{3}}}\)
(d) \({\rm{O}}_{\rm{2}}^{{\rm{2 - }}}\)
(e) \({{\rm{H}}_{\rm{2}}}{{\rm{O}}_{\rm{2}}}\)
