Chapter 7: Question 46 E (page 397)
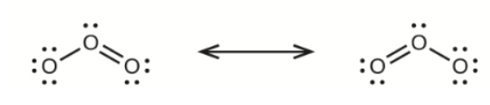
Write the resonance forms of ozone, \({{\rm{O}}_{\rm{3}}}\), the component of the upper atmosphere that protects the Earth from ultraviolet radiation.
Short Answer
The resonance forms of Ozone are-
Learning Materials
Features
Discover
Chapter 7: Question 46 E (page 397)
Write the resonance forms of ozone, \({{\rm{O}}_{\rm{3}}}\), the component of the upper atmosphere that protects the Earth from ultraviolet radiation.
The resonance forms of Ozone are-

All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the difference between a nonpolar covalent bond, a polar covalent bond, and an ionic bond.
Identify the molecules with a dipole moment:
Use the simulation (http://openstaxcollege.org/l/16MolecPolarity) to perform the following exercises for a two-atom molecule: (a) Adjust the electronegativity value so the bond dipole is pointing toward B. Then determine what the electronegativity values must be to switch the dipole so that it points toward A. (b) With a partial positive charge on A, turn on the electric field and describe what happens. (c) With a small partial negative charge on A, turn on the electric field and describe what happens. (d) Reset all, and then with a large partial negative charge on A, turn on the electric field and describe what happens.
Which of the following compounds requires the most energy to convert one mole of the solid into separate ions?
(a) \({\rm{MgO}}\)
(b) \({\rm{SrO}}\)
(c) \({\rm{KF}}\)
(d) \({\rm{CsF}}\)
(e) \({\rm{Mg}}{{\rm{F}}_{\rm{2}}}\)
Which of the following atoms would be expected to form negative ions in binary ionic compounds and which would be expected to form positive ions:\({\rm{P, I, Mg, Cl, In, Cs, O, Pb, Co}}\)?
What do you think about this solution?
We value your feedback to improve our textbook solutions.