Chapter 10: Q10.7P (page 405)
Draw a Lewis structure for (a); (b)(both H atoms are attached to O atoms); (c).
Short Answer
The Lewis structure of
a.
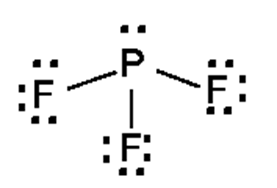
b.
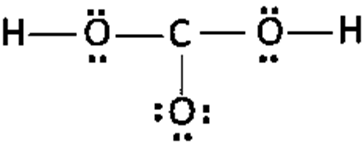
c.
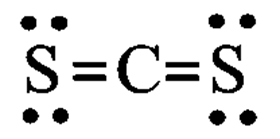
Learning Materials
Features
Discover
Chapter 10: Q10.7P (page 405)
Draw a Lewis structure for (a); (b)(both H atoms are attached to O atoms); (c).
The Lewis structure of
a.

b.

c.
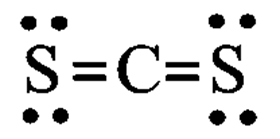
All the tools & learning materials you need for study success - in one app.
Get started for free
These species do not obey the octet rule. Draw a Lewis structure for each, and state the type of octet-rule exception:
(a)
(b)
(c)
Pure (atom sequence HNNN) is explosive. In aqueous solution, it is a weak acid that yields the azide ion, N. Draw resonance structures to explain why the nitrogen-nitrogen bond lengths are equal in but unequal in .
Draw Lewis structures of all the important resonance forms of
(a)
(b)
Determine the electron-group arrangement, molecular shape, and ideal bond angle(s) for each of the following:
(a)
(b)
(c)
Determine the shape, ideal bond angle(s), and the direction of any deviation from those angles for each of the following:
(a)
(b)
(c)
(d)
What do you think about this solution?
We value your feedback to improve our textbook solutions.