Chapter 10: Q10.11P (page 405)
Draw Lewis structures of all the important resonance forms of
(a)
(b)
Short Answer
The resonance hybrid form of
a.= Linear shape
b. = bent shape
Learning Materials
Features
Discover
Chapter 10: Q10.11P (page 405)
Draw Lewis structures of all the important resonance forms of
(a)
(b)
The resonance hybrid form of
a.= Linear shape
b. = bent shape
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw a Lewis structure for a resonance form of each ion with the lowest possible formal charges, show the charges, and give oxidation numbers of the atoms:
(a)
(b)
Draw a Lewis structure for (a); (b)(both H atoms are attached to O atoms); (c).
What would you expect to be the electron-group arrangement around atom A in each of the following cases? For each arrangement, give the ideal bond angle and the direction of any expected deviation:
Both aluminium and iodine form chlorides, and , with “bridging” Cl atoms. The Lewis structures are
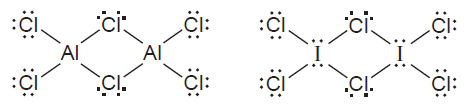
Hydrazine ( ) is used as a rocket fuel because it reacts very exothermically with oxygen to form nitrogen gas and water vapour. The heat released and the increase in the number of moles of gas provide thrust. Calculate the heat of the reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.