Chapter 3: Q8P (page 131)
3.8 Calculate the molar mass of each of the following:
(a) (b) (c) (d)
Short Answer
The molar mass of each of the following:
a)
b)
c)
d)
Learning Materials
Features
Discover
Chapter 3: Q8P (page 131)
3.8 Calculate the molar mass of each of the following:
(a) (b) (c) (d)
The molar mass of each of the following:
a)
b)
c)
d)
All the tools & learning materials you need for study success - in one app.
Get started for free
Each of the following balances weighs the indicated numbers of atoms of two elements:
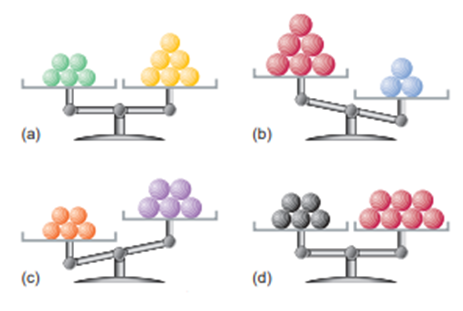
Which element-left, right, or neither,
(a) Has the higher molar mass?
(b) Has more atoms per gram?
(c) Has fewer atoms per gram?
(d) Has more atoms per mole?
Potassium nitrate decomposes on heating, producing potassium oxide and gaseous nitrogen and oxygen:
To produce 56.6 kg of oxygen, how many
(a) Moles of must be heated?
(b) Grams of must be heated?
Various nitrogen oxides, as well as sulfur oxides, contribute to acidic rainfall through complex reaction sequences. Nitrogen and oxygen combine during high-temperature combustion of fuels in air to form nitrogen monoxide gas, which reacts with more oxygen to form nitrogen dioxide gas. In contact with water vapor, nitrogen dioxide forms aqueous nitric acid and more nitrogen monoxide. (a) Write balanced equations for these reactions. (b) Use the equations to write one overall balanced equation that does not include nitrogen monoxide and nitrogen dioxide. (c) How many metric tons (t) of nitric acid form when 1350 t of atmospheric nitrogen is consumed (1 t = 1000 kg)?
Convert the following into balanced equations:
When nitrogen dioxide is bubbled into water, a solution ofnitric acid forms and gaseous nitrogen monoxide is released.
A chloride of silicon contains 79.1 mass % Cl.
(a) What is the empirical formula of the chloride?
(b) If the molar mass is 269 g/mol, what is the molecular formula?
What do you think about this solution?
We value your feedback to improve our textbook solutions.