Chapter 3: 3.53P (page 133)
Write balanced equations for each of the following by inserting the correct coefficients in the blanks:
Learning Materials
Features
Discover
Chapter 3: 3.53P (page 133)
Write balanced equations for each of the following by inserting the correct coefficients in the blanks:
All the tools & learning materials you need for study success - in one app.
Get started for free
Hemoglobin is 6.0% heme (C34H32FeN4O4) by mass. To remove the heme, hemoglobin is treated with acetic acid and NaCl to form hemin (C34H32N4O4FeCl). At a crime scene, a blood sample contains 0.65 g of hemoglobin. (a) How many grams of heme are in the sample? (b) How many moles of heme? (c) How many grams of Fe? (d) How many grams of hemin could be formed for a forensic chemist to measure?
Each of the following balances weighs the indicated numbers of atoms of two elements:
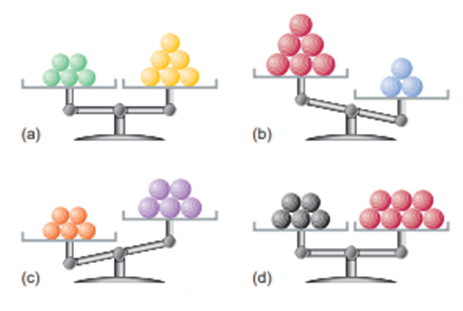
Which element-left, right, or neither,
(a) Has the higher molar mass?
(b) Has more atoms per gram?
(c) Has fewer atoms per gram?
(d) Has more atoms per mole?
Question. In the process of balancing the equation
Student I writes:
Student II writes:
Student III writes:
Is the approach of Student I valid? Student II? Student III? Explain.
Cocaine (C17H21O4N) is a natural substance found in coca leaves, which have been used for centuries as a local anesthetic and stimulant. Illegal cocaine arrives in the United States either as the pure compound or as the hydrochloride salt (C17H21O4NHCl). At 25oC, the salt is very soluble in water (2.50 kg/L), but cocaine is much less so (1.70 g/L). (a) What is the maximum amount (in g) of the salt that can dissolve in 50.0 mL of water? (b) If the solution in part (a) is treated with NaOH, the salt is converted to cocaine. How much additional water (in L) is needed to dissolve it?
Calculate each of the following quantities:
(a) Mass in grams of 0.68 mol of
(b) Moles of O atoms in 8.18 g of
(c) Number of O atoms in of
What do you think about this solution?
We value your feedback to improve our textbook solutions.