Chapter 12: Q12.51P (page 495)
Which substance has the lower boiling point? Explain.
a.OR
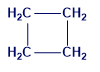
b.
c.
Short Answer
a.
b.
c.
Learning Materials
Features
Discover
Chapter 12: Q12.51P (page 495)
Which substance has the lower boiling point? Explain.
a.OR

b.
c.
a.
b.
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the effect (if any) of a decrease in temperature on the electrical conductivity of (a) silicon; (b) lead; (c) germanium.
An element crystallizes in a face-centered cubic lattice and has a density of . The edge of its unit cell is 4.52108 cm.
(a) How many atoms are in each unit cell?
(b) What is the volume of a unit cell?
(c) What is the mass of a unit cell?
(d) Calculate an approximate atomic mass for the element.
When benzene is at its melting point, two processes occur simultaneously and balance each other. Describe these processes on the macroscopic and molecular levels.
Sulfur dioxide is produced in enormous amounts for sulfuric acid production. It melts at and boils at . Its is 8.619 kJ/mol and itsis 25.73 kJ/mol. The specific heat capacities of the liquid and gas are 0.995 J/gK and 0.622 J/gK, respectively. How much heat is required to convert 2.500 kg of solid role="math" localid="1656937400249" at the melting point to a gas at ?
Why are covalent bonds typically much stronger than intermolecular forces?
What do you think about this solution?
We value your feedback to improve our textbook solutions.