Chapter 12: Q12.52P (page 495)
Which substance has the higher boiling point? Explain.
a.OR
b. OR
c.
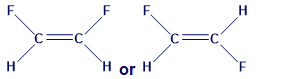
Short Answer
The following substance have higher boiling point:
Learning Materials
Features
Discover
Chapter 12: Q12.52P (page 495)
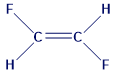
Which substance has the higher boiling point? Explain.
a.OR
b. OR
c.

The following substance have higher boiling point:

All the tools & learning materials you need for study success - in one app.
Get started for free
Use these data to draw a qualitative phase diagram for . Does sublime at 0.05atm? Explain.
MP at 1atm: 13.96 K
BP at 1atm: 20.39 K
Triple point: 13.95 K and 0.07atm
Critical point: 33.2 K and 13.0atm
Vapour pressure of solid at 10 K: 0.001atm
What is the strongest inter-particle force in each substance?
(a) (b) (c)
Which forces are intramolecular and which intermolecular?
(a) Those preventing oil from evaporating at room temperature
(b) Those preventing butter from melting in a refrigerator
(c) Those allowing the silver to tarnish
(d) Those preventing in the air from forming O atoms
Why are covalent bonds typically much stronger than intermolecular forces?
Which forces oppose the vaporization of each substance?
(a), (b) data-custom-editor="chemistry" , (c)data-custom-editor="chemistry"
What do you think about this solution?
We value your feedback to improve our textbook solutions.