Chapter 12: 12.105P (page 497)
Distinguish between isotropic and anisotropic substances. To which category do liquid crystals belong?
Short Answer
Liquid crystal flowslike a liquid and has crystalline structure.
Learning Materials
Features
Discover
Chapter 12: 12.105P (page 497)
Distinguish between isotropic and anisotropic substances. To which category do liquid crystals belong?
Liquid crystal flowslike a liquid and has crystalline structure.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: What type of forces, intramolecular or intermolecular:
Which member in each pair of liquids has the higher vapour pressure at a given temperature? Explain.
(a) or (b) or (c) or
In the process of doping a semiconductor, certain impurities are added to increase its electrical conductivity. Explain this process for an n-type and a p-type semiconductor.
Polonium, the Period 6 member of Group 6A (16), is a rare radioactive metal that is the only element with a crystal structure based on the simple cubic unit cell. If its density is, calculate an approximate atomic radius for polonium.
Intermolecular forces are depicted in the scenes below:
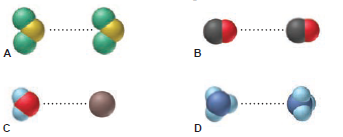
(a) Name the type of force being depicted in each scene.
(b) Rank the forces in order of increasing strength.
What do you think about this solution?
We value your feedback to improve our textbook solutions.