Chapter 21: Q21.6P (page 972)
How are protons removed when balancing a redox reaction in a basic solution?
Short Answer
By introducing an equal number of on both sides of the reaction, the resultant ions are balanced.
Learning Materials
Features
Discover
Chapter 21: Q21.6P (page 972)
How are protons removed when balancing a redox reaction in a basic solution?
By introducing an equal number of on both sides of the reaction, the resultant ions are balanced.
All the tools & learning materials you need for study success - in one app.
Get started for free
What are and of a redox reaction at for which and?
Consider the following general electrolytic cell:
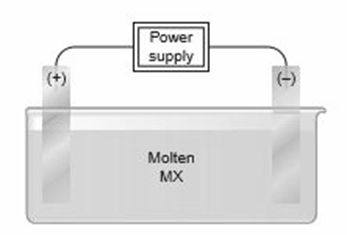
(a) At which electrode does oxidation occur?
(b) At which electrode does elemental M form?
(c) At which electrode are electrons being released by ions?
(d) At which electrode are electrons entering the cell?
When a clean iron nail is placed in an aqueous solution of copper(II) sulfate, the nail becomes coated with a brownish black material.
(a) What is the material coating the iron?
(b) What are the oxidizing and reducing agents?
(c) Can this reaction be made into a voltaic cell?
(d) Write the balanced equation for the reaction.
(e) Calculate E°cell for the process.
A concentration cell consists of two half-cells. The electrolyte in compartment A is 0.13 M . The electrolyte in B is 0.87M . Which half-cell houses the cathode? What is the voltage of the cell?
A voltaic cell is constructed with anhalf-cell and ahalf-cell. The zinc electrode is negative.
(a) Write balanced half-reactions and the overall reaction.
(b) Diagram the cell, labeling electrodes with their charges and showing the directions of electron flow in the circuit and of cation and anion flow in the salt bridge.
What do you think about this solution?
We value your feedback to improve our textbook solutions.