Chapter 21: Q21.67P (page 922)
What are and of a redox reaction atfor which and?
Learning Materials
Features
Discover
Chapter 21: Q21.67P (page 922)
What are and of a redox reaction atfor which and?
All the tools & learning materials you need for study success - in one app.
Get started for free
Consider the following voltaic cell
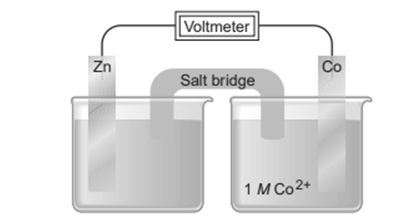
(a) In which direction do electrons flow in the external circuit?
(b) In which half-cell does oxidation occur?
(c) In which half-cell do electrons enter the cell?
(d) At which electrode are electrons consumed?
(e) Which electrode is negatively charged?
(f) Which electrode decreases in mass during cell operation?
(g) Suggest a solution for the cathode electrolyte.
(h) Suggest a pair of ions for the salt bridge.
(i) For which electrode could you use an inactive material?
(j) In which direction do anions within the salt bridge move to maintain charge neutrality?
(k) Write balanced half-reactions and an overall cell reaction.
What purpose does the salt bridge serve in a voltaic cell, and how does it accomplish this purpose?
The standard cell potential is a thermodynamic state function. How arevalues treated similarly tovalues? How are they treated differently?
The overall cell reaction occurring in an alkaline battery is
(a) How many moles of electrons flow per mole of reaction?
(b) Ifof zinc is oxidized, how many grams of manganese dioxide and of water are consumed?
(c) What is the total mass of reactants consumed in part (b)?
(d) How many coulombs are produced in part (b)?
(e) In practice, voltaic cells of a given capacity (coulombs) are heavier than the calculation in part (c) indicates. Explain.
What are and of a redox reaction at for which and?
What do you think about this solution?
We value your feedback to improve our textbook solutions.