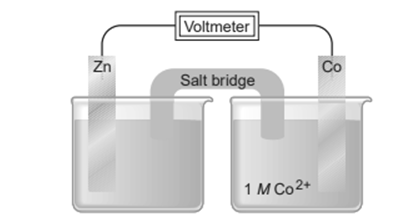
Chapter 21: Q21.112Cp (page 922)
The overall cell reaction occurring in an alkaline battery is
(a) How many moles of electrons flow per mole of reaction?
(b) Ifof zinc is oxidized, how many grams of manganese dioxide and of water are consumed?
(c) What is the total mass of reactants consumed in part (b)?
(d) How many coulombs are produced in part (b)?
(e) In practice, voltaic cells of a given capacity (coulombs) are heavier than the calculation in part (c) indicates. Explain.
Short Answer
(a) The number of moles of electrons that flow per mole of reaction is 2 moles. .
(b) The amount of manganese dioxide and water that are consumed is and respectively.
(c) The total mass of reactants consumed is .
(d) The charged produced is .
(e) Batteries are heavier because they have a steel container, outer jacket, brass pin current collector, separator, steal vent, and metal base in addition to the cathode and anode.