Chapter 21: Q21.31 P (page 973)
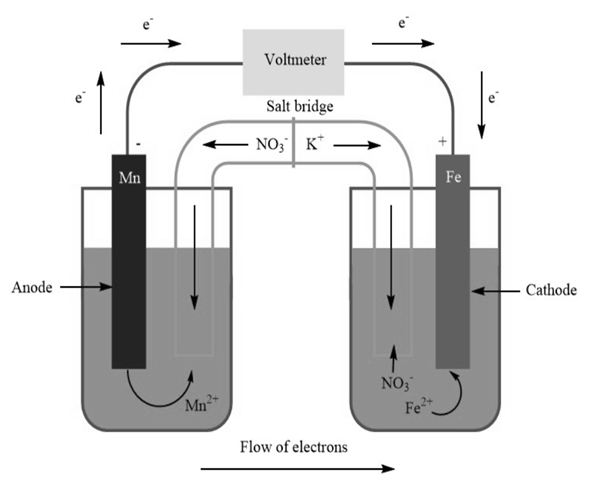
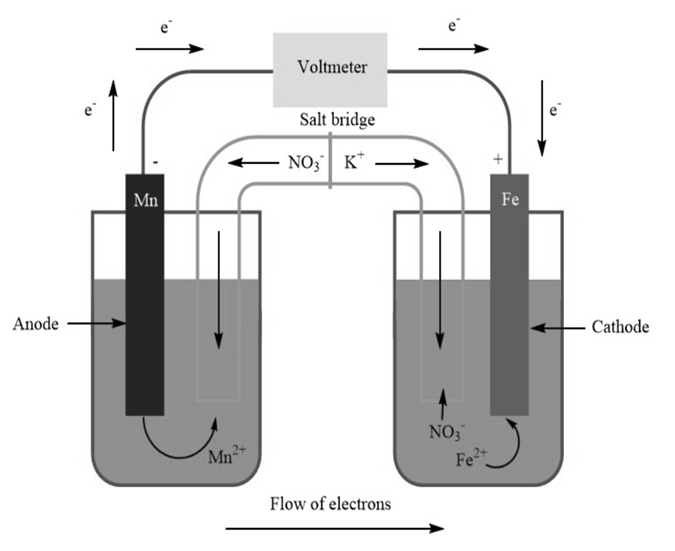
A voltaic cell is constructed with an half-cell and an half-cell. The iron electrode is positive.
(a) Write balanced half-reactions and the overall reaction.
(b) Diagram the cell, labeling electrodes with their charges and showing the directions of electron flow in the circuit and of cation and anion flow in the salt bridge.
Short Answer
a) Overall reaction
b)