Chapter 18: Q39P (page 824)
(a) A Brønsted-Lowry acid-base reaction proceeds in the net direction in which a stronger acid and stronger base form a weaker acid and weaker base. Explain
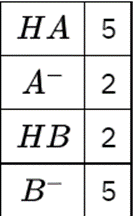
(b) The molecular scene at the right depicts an aqueous solution of two conjugate acid-base pairs: HA/A and HB/B. The base in the first pair is represented by red spheres and the base in the second pair by green spheres; solvent molecules are omitted for clarity. Which is the stronger acid? Stronger base? Explain.

Short Answer
(a) Strong acids and bases dissociate, whereas weak acids and bases dissociate only partially.
(b) is the stronger base and HB is the stronger acid