Chapter 9: Q. 9.48 (page 306)
Calculate the grams of solute needed to prepare each of the following:
a. solution
b. solution
c. solution
Short Answer
a. The grams of is.
b. The grams of is.
c. The grams of is.
Learning Materials
Features
Discover
Chapter 9: Q. 9.48 (page 306)
Calculate the grams of solute needed to prepare each of the following:
a. solution
b. solution
c. solution
a. The grams of is.
b. The grams of is.
c. The grams of is.
All the tools & learning materials you need for study success - in one app.
Get started for free
Explain the following observations:
a. More sugar dissolves in hot tea than in iced tea.
b. Champagne in a warm room goes flat.
c. A warm can of soda has more spray when opened than a cold one.
How many liters of a solution would you need to obtain ?
Describe the formation of an aqueous solution, when solid dissolves in water.
If all the solute is dissolved in diagram , how would heating or cooling the solution cause each of the following changes? (9.3)
a.
b.
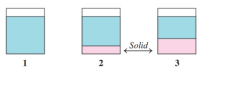
How do temperature and pressure affect the solubility of solids and gases in water?
What do you think about this solution?
We value your feedback to improve our textbook solutions.