Chapter 9: Q. 9.4 (page 289)
Describe the formation of an aqueous solution, when solid dissolves in water.
Short Answer
If ions like and dissolved in the water, the water particles in attract the ions and pull them into solution.
Learning Materials
Features
Discover
Chapter 9: Q. 9.4 (page 289)
Describe the formation of an aqueous solution, when solid dissolves in water.
If ions like and dissolved in the water, the water particles in attract the ions and pull them into solution.
All the tools & learning materials you need for study success - in one app.
Get started for free
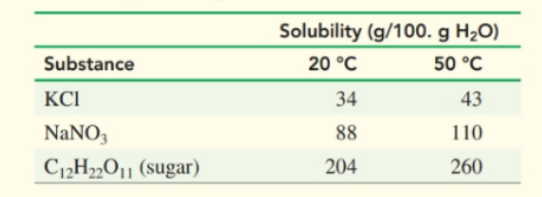
A solution containing in at is cooled to
a. How many grams of remain in solution at ?
b. How many grams of solid crystallized after cooling?
Why does iodine dissolve in hexane, but not in water?
If the solid in a saturated solution of continues to dissolve, why is there no change in the concentration of the solution?
What is the initial volume, in milliliters, needed to prepare each of the following diluted solutions?
a. of % from %
b. . of % role="math" localid="1652626186960" from % role="math" localid="1652626200339"
c. . of from
What is the initial volume, in milliliters, needed to prepare each of the following diluted solutions? (9.5)
a. of glucose from glucose
b. of from
c.offrom
What do you think about this solution?
We value your feedback to improve our textbook solutions.