Chapter 9: Q. 9.102 (page 318)
How many milliliters of a solution contain of
Short Answer
of solution of solution contains of
Learning Materials
Features
Discover
Chapter 9: Q. 9.102 (page 318)
How many milliliters of a solution contain of
of solution of solution contains of
All the tools & learning materials you need for study success - in one app.
Get started for free
Indicate whether aqueous solutions of each of the following solutes contain only ions, only molecules, or mostly molecules and a few ions:
a. , a strong electrolyte.
b. ethanol, , a nonelectrolyte.
c., hydrocyanic acid, a weak electrolyte.
How do temperature and pressure affect the solubility of solids and gases in water?
Determine whether each of the following solutions will be saturated or unsaturated at :
a. adding of to role="math" localid="1652588032291"
b. adding to role="math" localid="1652588024461"
c. adding of sugar to
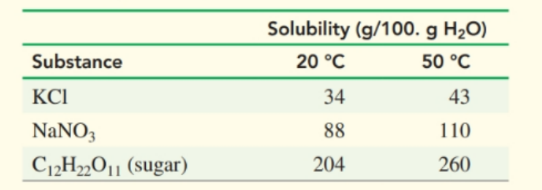
Select the diagram that represents the solution formed by a solute that is a
a. nonelectrolyte
b. weak electrolyte
c. strong electrolyte

A patient needs of glucose in the next . How many liters of a glucose solution must be given?
What do you think about this solution?
We value your feedback to improve our textbook solutions.