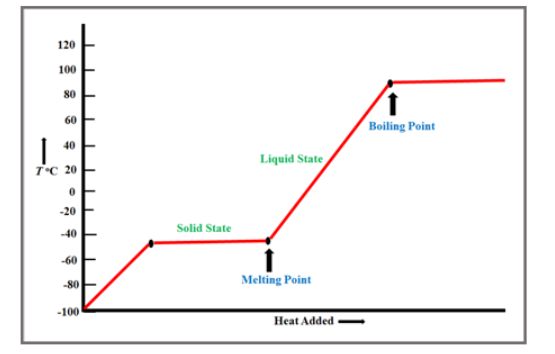
Chapter 3: Q.3.91 (page 93)
The melting point of dibromomethane is and its boiling point is . Sketch a heating curve for dibromomethane from .
a. What is the state of dibromomethane at ?
b. What happens on the curve at ?
c. What is the state of dibromomethane at ?
d. What is the state of dibromomethane at ?
e. At what temperature will both solid and liquid be present?
Short Answer
a. dibromomethane is in solid-state
b. It will be parallel to the x-axis.
c. dibromomethane is in a liquid state
d. dibromomethane is in a gaseous state
e. At . Its melting point.
The graph is shown