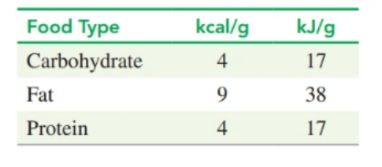
Using the energy values for foods (see Table), determine each of the following (round off the answer for each food type to the tens place):
(a) the total kilojoules for one cup of orange juice that contains 26 g of carbohydrate, no fat, and of protein
(b) the grams of carbohydrate in one apple if the apple has no fat and no protein and provides of energy
(c) the kilocalories in one tablespoon of vegetable oil, which containsof fat and no carbohydrate or protein
(d )the grams of fat in one avocado that has , of carbohydrate, and of protein