Chapter 3: Q. 3.34 (page 77)
For lunch, Charles consumed of skinless chicken, of broccoli, medium apple, and cup of nonfat milk (see Table 3.8). How many kilocalories did Charles obtain from the lunch?
Short Answer
The amont of energy from lunch is
Learning Materials
Features
Discover
Chapter 3: Q. 3.34 (page 77)
For lunch, Charles consumed of skinless chicken, of broccoli, medium apple, and cup of nonfat milk (see Table 3.8). How many kilocalories did Charles obtain from the lunch?
The amont of energy from lunch is
All the tools & learning materials you need for study success - in one app.
Get started for free
Compost can be made at home from grass clippings, kitchen scraps, and dry leaves. As microbes break down organic matter, heat is generated and the compost can reach a temperature of , which kills most pathogens. What is this temperature in degrees Celsius? In kelvins? (3.3)

Compost produced from decayed plant material is used to enrich the soil.
In one box of nails, there are iron nails weighing . The density of iron is . The specific heat of iron is . The melting point of iron is ..
What is the volume, in cubic centimeters, of the iron nails in the box?
If 30 nails are added to a graduated cylinder containing of water, what is the new level of water, in milliliters, in the cylinder?
How much heat, in joules, must be added to the nails in the box to raise their temperature from to ?
How much heat, in joules, is required to heat one nail from to its melting point?

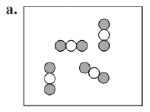
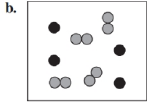
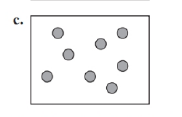
Identify each of the following as an element, a compound, or a mixture. Explain your choice. (3.1)
If you want to lose of "body fat," which is water, how many kilocalories do you need to expend?
Using the values for the heat of fusion, specific heat of water. and/or heat of vaporization, calculate the amount of heat energy in each of the following:
a. joules released when of steam at condenses and cools to liquid at
b. kilocalories needed to melt a ice sculpture at and to warm the liquid to
c. kilojoules released when of steam condenses at , cools, and freezes at
What do you think about this solution?
We value your feedback to improve our textbook solutions.