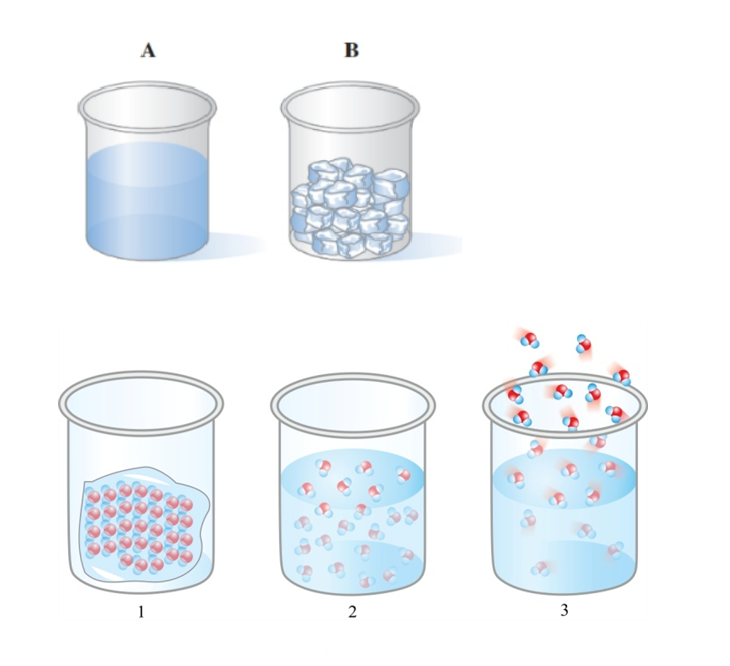
Chapter 3: Q. 3.65 (page 91)
Compost can be made at home from grass clippings, kitchen scraps, and dry leaves. As microbes break down organic matter, heat is generated and the compost can reach a temperature of , which kills most pathogens. What is this temperature in degrees Celsius? In kelvins? (3.3)

Compost produced from decayed plant material is used to enrich the soil.
Short Answer
Therefore the temperature in Celsius is and in Kelvin is