Chapter 3: Q. 3.64 (page 91)
State the temperature on the Celsius thermometer and convert to Fahrenheit.
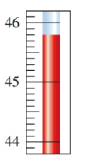
Short Answer
Therefore,is the temperature.
Learning Materials
Features
Discover
Chapter 3: Q. 3.64 (page 91)
State the temperature on the Celsius thermometer and convert to Fahrenheit.

Therefore,is the temperature.
All the tools & learning materials you need for study success - in one app.
Get started for free
Oa a hot sunny day, you get out of the swimming pool and sit in a metal chair, which is very hot. Would you predict that the specific heat of the metal is higher or lower than that of water? Explain.
Classify each of the following as a pure substance or a mixture:
a. a soft drink
b. propane
c. a cheese sandwich
d. an iron () nail
e. salt substitutelocalid="1651731802252"
Using the values for the heat of fusion, specific heat of water, and/or heat of vaporization, calculate the amount of heat energy in each of the following:
a. joules needed to melt of ice at and to warm the liquid to
b. Kilocalories released when of steam condenses at and the liquid cools to
C. kilojoules needed to melt of ice at , warm the liquid to , and change it to steam at .
Discuss the changes in the potential and kinetic energy of a ski jumper taking the elevator to the top of the jump and going down the ramp.
In one box of nails, there are iron nails weighing . The density of iron is . The specific heat of iron is . The melting point of iron is ..
What is the volume, in cubic centimeters, of the iron nails in the box?
If 30 nails are added to a graduated cylinder containing of water, what is the new level of water, in milliliters, in the cylinder?
How much heat, in joules, must be added to the nails in the box to raise their temperature from to ?
How much heat, in joules, is required to heat one nail from to its melting point?

What do you think about this solution?
We value your feedback to improve our textbook solutions.