Chapter 3: Q. 3.88 (page 93)
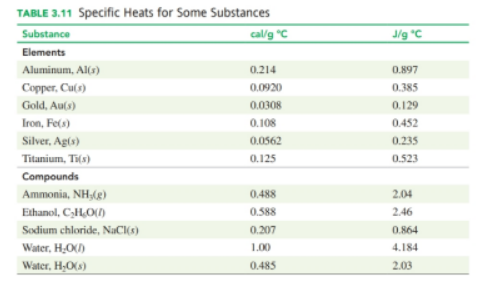
Oa a hot sunny day, you get out of the swimming pool and sit in a metal chair, which is very hot. Would you predict that the specific heat of the metal is higher or lower than that of water? Explain.
Short Answer
As a result, the temperature of a metal chair rises faster than that of water