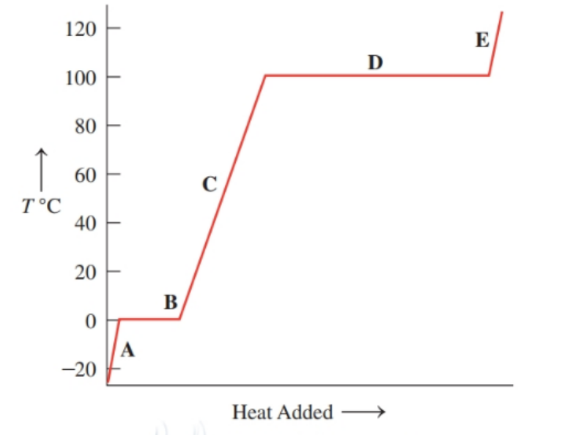
Chapter 3: Q.3.39 (page 80)
Use the heat equation to calculate the energy for each of the following (see Table 3.11 ):
a. calories to heat 8.5g of water from
b. joules lost when 25g of water cools from
c. kilocalories to heat 150g of water from
d. kilojoules to heat 175 g of copper from
Short Answer
a.
b.
c.
d.