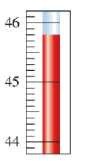
Chapter 3: Q. 3.97 (page 94)
When a sample of olive oil is burned in a calorimeter, the heat released increases the temperature of of water from to . What is the energy value for the olive oil in sample?
Short Answer
The energy value of olive oil arein Kilojoule andin Kilocalories.