Chapter 14: Q. 14.16 (page 481)
Draw the condensed structural formula for the ester formed when each of the following reacts with ethyl alcohol:
a. formic acid
b. propionic acid
Short Answer
(part a) 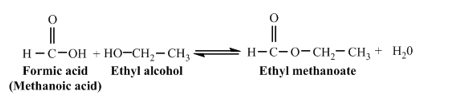
(part b) 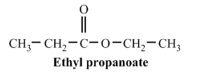
Learning Materials
Features
Discover
Chapter 14: Q. 14.16 (page 481)
Draw the condensed structural formula for the ester formed when each of the following reacts with ethyl alcohol:
a. formic acid
b. propionic acid
(part a) 
(part b) 
All the tools & learning materials you need for study success - in one app.
Get started for free
4 Draw the condensed structural or line-angle formulas for the products from the hydrolysis of each of the following: (14.6)
Identify the compound in each group that is most soluble in water. Explain.
a. propanoic acid, hexanoic acid, benzō̄ic acid
b. pentane, -hexanol, propanoic acid
Write the balanced chemical equation for the dissociation of each of the following carboxylic acids in water:
a. pentanoic acid
b. acetic acid
Classify each of the following amines as primary , secondary , or tertiary :
a.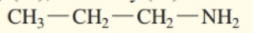
b.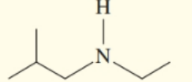
c. 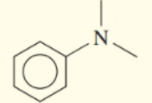
d. 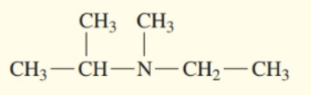
Draw the condensed structural or line-angle formula for the ester formed in each of the following reactions:
(a) 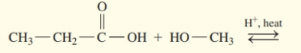
(b) 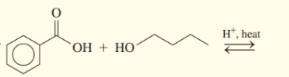
What do you think about this solution?
We value your feedback to improve our textbook solutions.