Chapter 14: Q. 14.18 (page 482)
Draw the condensed structural or line-angle formula for the ester formed in each of the following reactions:
(a) 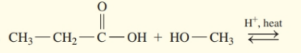
(b) 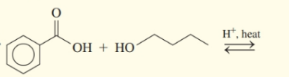
Short Answer
(part a) 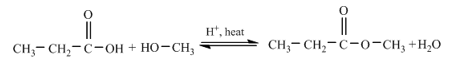
(part b) 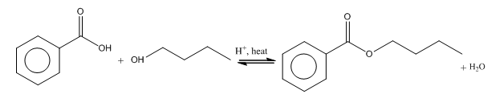
Learning Materials
Features
Discover
Chapter 14: Q. 14.18 (page 482)
Draw the condensed structural or line-angle formula for the ester formed in each of the following reactions:
(a) 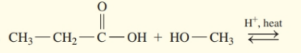
(b) 
(part a) 
(part b) 
All the tools & learning materials you need for study success - in one app.
Get started for free
Draw the condensed structural or line-angle formulas for the products of the following:
a)
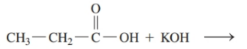
b)
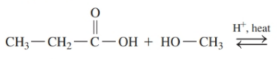
c)
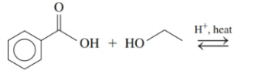
d)
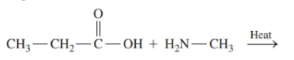
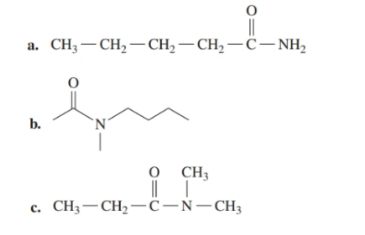
Write the IUPAC name for each of the following:
a. 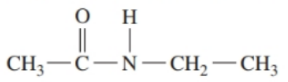
b. 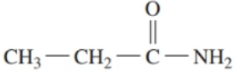
c. 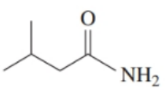
Write the IUPAC name for each of the following: (14.6)
Propyl acetate is the ester that gives the odor and smell of pears.
a. Draw the condensed structural formula for propyl acetate.
b. Write the balanced chemical equation for the formation of propyl acetate.
c. Write the balanced chemical equation for the acid hydrolysis of propyl acetate.
d. Write the balanced chemical equation for the base hydrolysis of propyl acetate with .
e. How many milliliters of a solution is needed to completely hydrolyze (saponify)
of propyl acetate?
Write the balanced chemical equations for the reaction of each of the following amines with water and neutralization with HCl:
a. methylamine
b. dimethylamine
c. aniline
What do you think about this solution?
We value your feedback to improve our textbook solutions.