Chapter 10: Q. 72 (page 358)
Identify each of the following as an acid or a base; (10.1)
A.
B.
C.
D.
Short Answer
(Part a). is an strong base.
(Part b). is a weak acid
(Part c). is a weak acid
(Part d). is an strong base.
Learning Materials
Features
Discover
Chapter 10: Q. 72 (page 358)
Identify each of the following as an acid or a base; (10.1)
A.
B.
C.
D.
(Part a). is an strong base.
(Part b). is a weak acid
(Part c). is a weak acid
(Part d). is an strong base.
All the tools & learning materials you need for study success - in one app.
Get started for free
How many grams of are required to neutralize 100mL of stomach acid, which is equivalent to 0.0400 M HCL?
If of a solution is required to titrate of a solution of , what is the molarity of the solution?
If you know the , how can you determine the pH of a solution?
Complete the following table-
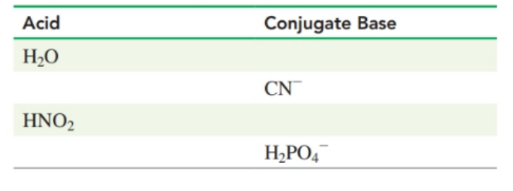
Identify the Brønsted-Lowry acid-base pairs in each of the following equations:
What do you think about this solution?
We value your feedback to improve our textbook solutions.