Chapter 10: Q.10.73 (page 358)
Complete the following table-
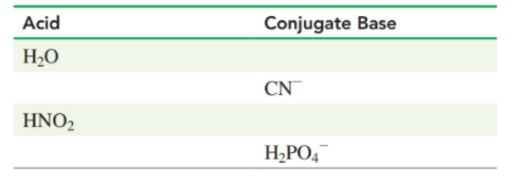
Short Answer
conjugate base-
acid- HCN
conjugate base- role="math" localid="1653111199726"
acid-
Learning Materials
Features
Discover
Chapter 10: Q.10.73 (page 358)
Complete the following table-

conjugate base-
acid- HCN
conjugate base- role="math" localid="1653111199726"
acid-
All the tools & learning materials you need for study success - in one app.
Get started for free
10.25 Why are the concentrations ofandequal in pure water?
What is meant by the term reversible reaction?
Calculate the volume, in millilitres, of a 0.215 M KOH solution that will completely neutralize each of the following:
a. 2.50 mL of asolution
b. 18.5 mL of a solution
c. 5.00 mL of asolution
Identify the stronger acid in each of the following pairs: (10.3)
a. HF or HCN
b.
c.
d.
Write the formula for the conjugate acid for each of the following bases:
a.
b.
c.
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.