Chapter 24: Q6CP (page 847)
Why is it necessary for naturally occurring DNA molecules to be negatively supercoiled?
Short Answer
The negative supercoiling prepares the molecule for the molecules that need separation of the DNA strands.
Learning Materials
Features
Discover
Chapter 24: Q6CP (page 847)
Why is it necessary for naturally occurring DNA molecules to be negatively supercoiled?
The negative supercoiling prepares the molecule for the molecules that need separation of the DNA strands.
All the tools & learning materials you need for study success - in one app.
Get started for free
The asterisk in the structure shown in Problem 31 indicates the site where the base is attached via a two-carbon linker. Compare the overall backbone length for a PNA and a DNA molecule and explain why the two molecules can form base-paired duplexes.
Which of the DNAs shown in Figure 24-8 would move fastest during agarose gel electrophoresis?
Why does it make metabolic sense for UTP to inhibit carbamoylphosphate synthetase II, whereas ATP activates the enzyme?
The melting curve for the polyribonucleotide poly(A) is shown below.
(a) Explain why absorbance increases with increasing temperature.
(b) Why does the shape of the curve differ from the one shown in Fig. 24-22?
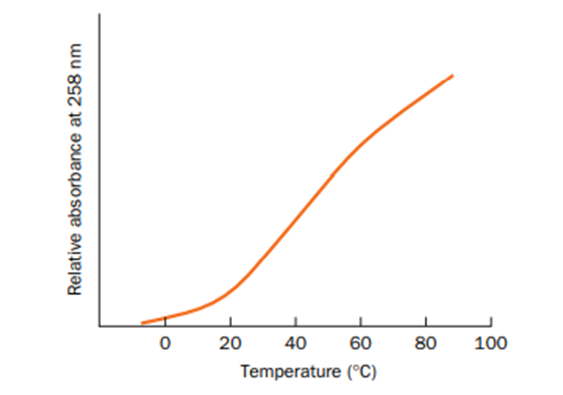
How is the melting curve of duplex DNA affected by decreasing the ionic strength of the solution?
What do you think about this solution?
We value your feedback to improve our textbook solutions.