Chapter 14: Q17P (page 476)
Under standard conditions, will the following reaction proceed spontaneously as written?
Short Answer
The reaction will unfold spontaneously as written.
Learning Materials
Features
Discover
Chapter 14: Q17P (page 476)
Under standard conditions, will the following reaction proceed spontaneously as written?
The reaction will unfold spontaneously as written.
All the tools & learning materials you need for study success - in one app.
Get started for free
How is Δℰrelated to ΔG?
Assuming 100% efficiency of energy conservation, how many moles of ATP can be synthesized under standard conditions by the complete oxidation of 1 mol of glucose?
In the partial reactions shown below, is the reactant undergoing oxidation or reduction?
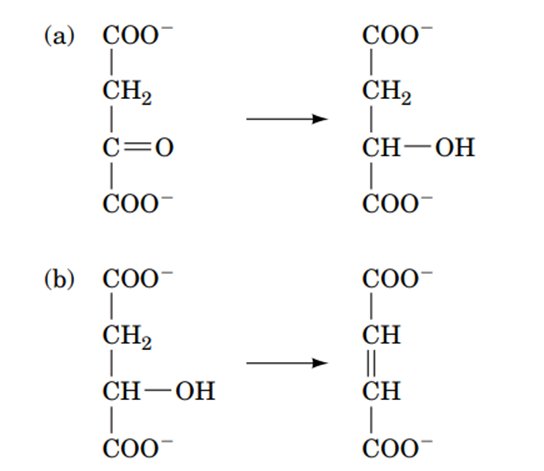
Question:Cells carry out anabolic as well as catabolic pathways, with some enzymes functioning in both types of pathways. (a) Explain why these enzymes catalyze near-equilibrium reactions. (b) Explain why opposing anabolic and catabolic pathways must have different enzymes for at least one of the steps.
Question: Is the reduced form of cytochrome c more likely to give up its electron to oxidized cytochrome a or cytochrome b?
What do you think about this solution?
We value your feedback to improve our textbook solutions.