Chapter 12: Q7. (page 373)
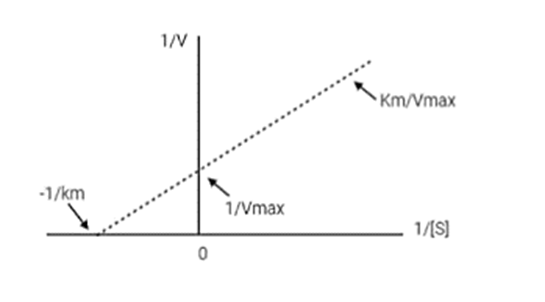
Write the Lineweaver–Burk (double reciprocal) equation and describe the features of a Lineweaver–Burk plot.
Short Answer
The double reciprocal equation of the Lineweaver Burk plot is:
The main feature is that it provides a more precise estimation of on inhibition.