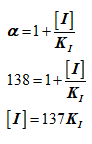Chapter 12: Q31P (page 400)
Ethanol in the body is oxidized to acetaldehyde (CH₃CHO) by liveralcohol dehydrogenase (LADH). Other alcohols are also oxidized byLADH. For example, methanol CH₃OH), which is mildly intoxicating, isoxidized by LADH to the quite toxic product formaldehyde (CH₂O). Thetoxic effects of ingesting methanol (a component of many commercialsolvents) can be reduced by administering ethanol. The ethanol acts as acompetitive inhibitor of the methanol by displacing it from LADH. Thisprovides sufficient time for the methanol to be harmlessly excreted bythe kidneys. If an individual has ingested 100 mL of methanol (a lethaldose), how much 100 proof whiskey (50% ethanol by volume) must beimbibe to reduce the activity of his LADH toward methanol to 5% of itsoriginal value? The adult human body contains -40 L of aqueous fluidsthroughout which ingested alcohols are rapidly and uniformly mixed.The densities of ethanol and methanol are both 0.79 g .cm-3. Assumethe KM values of LADH for ethanol and methanol to be 1.0 X 10⁻³Mand 1.0 X 10⁻²M, respectively, and that KI = KM for ethanol.
Short Answer
The quantity of 100 proof whiskeys a person who has ingested 100mL ethanol must absorb to reduce the activity of liver alcohol dehydrogenase is 638mL.