Chapter 11: Q17P (page 359)
Design a chloromethylketone inhibitor of elastase.
Short Answer
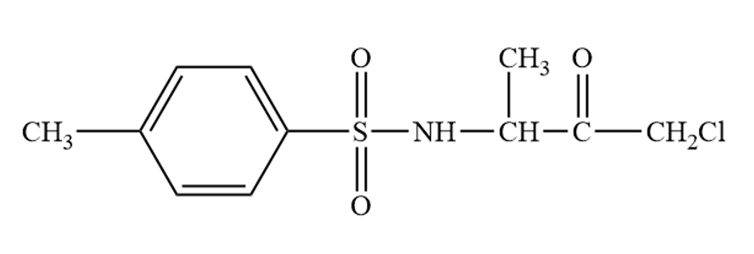
Tosyl-L-alanine chloromethylkeyone
Learning Materials
Features
Discover
Chapter 11: Q17P (page 359)
Design a chloromethylketone inhibitor of elastase.

Tosyl-L-alanine chloromethylkeyone
All the tools & learning materials you need for study success - in one app.
Get started for free
Predict the effect on lysozyme’s activity of mutating Glu 35 to Asp and Asp 52 to Glu.
Question: Predict the effect of mutating Asp 102 of trypsin to Asn (a) on substrate binding and (b) on catalysis.
Diagram the hydrogen-bonding interactions of the catalytic triad His–Lys–Ser during catalysis in a hypothetical hydrolytic enzyme.
Lysozyme residues Asp 101 and Arg 114 are required for efficient catalysis, although they are located some distance from the active sites Glu 35 and Asp 52. Substituting Ala for either Asp 101 or Arg 114 does not significantly alter the enzyme’s tertiary structure, but it significantly reduces its catalytic activity. Explain.
Why are cofactors required for some enzymatic reactions?
What do you think about this solution?
We value your feedback to improve our textbook solutions.