Chapter 4: Q5P (page 95)
Draw the dipeptide Asp-His at pH 7.0
Short Answer
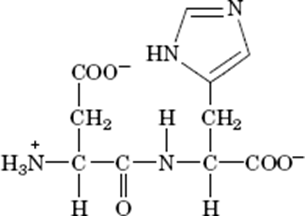
The dipeptide Asp-His at pH 7.0 is as follows –
Learning Materials
Features
Discover
Chapter 4: Q5P (page 95)
Draw the dipeptide Asp-His at pH 7.0
The dipeptide Asp-His at pH 7.0 is as follows –

All the tools & learning materials you need for study success - in one app.
Get started for free
Estimate the isoelectric point of a Ser–His dipeptide. Explain why this value is only an estimate.
A sample of the amino acid tyrosine is barely soluble in water. Would a polypeptide containing only Tyr residues, poly(Tyr), be more or less soluble, assuming the total number of Tyr groups remains constant?
Why do the values of ionizable groups differ between free amino acids and amino acid residues in polypeptides?
Identify the hydrogen bond donor and acceptor groups in asparagine.
Identify the amino acids that differ from each other by a single methylor methylene group.
What do you think about this solution?
We value your feedback to improve our textbook solutions.