Chapter 2: Q2.2-2ITD (page 33)
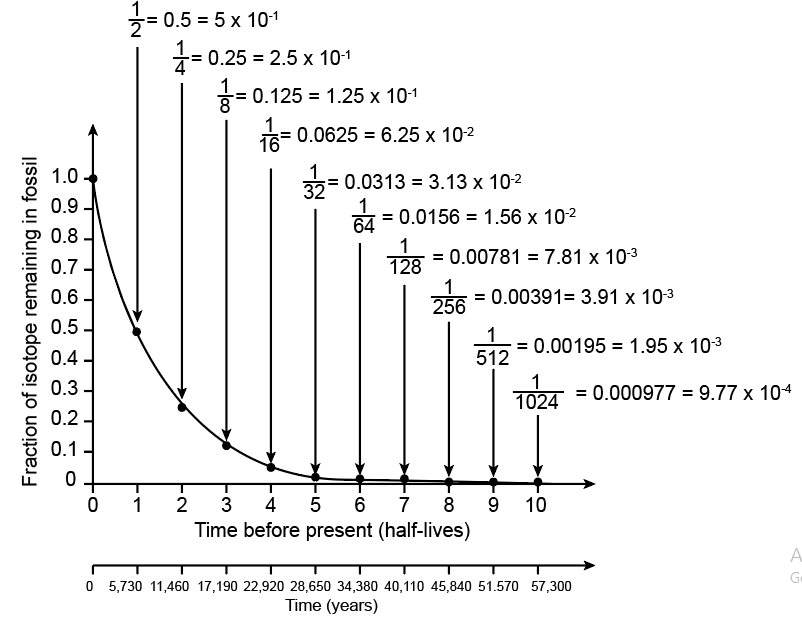
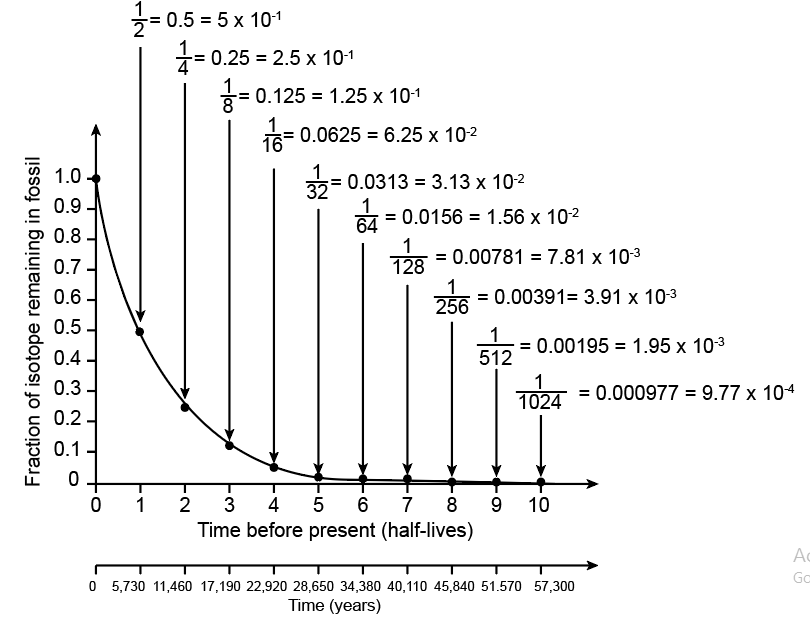
Recall that 14C has a half life of 5,730 years. To calibrate the x-axis for 14C decay, write the time before present in years below each half-life.
Short Answer
Learning Materials
Features
Discover
Chapter 2: Q2.2-2ITD (page 33)
Recall that 14C has a half life of 5,730 years. To calibrate the x-axis for 14C decay, write the time before present in years below each half-life.

All the tools & learning materials you need for study success - in one app.
Get started for free
If you were a pharmaceutical researcher, why would you want to learn the three-dimensional shapes of naturally occurring signaling molecules?
Why does the structure H-C=C-H fail to make sense chemically?
We can represent atoms by listing the number of protons, neutrons, and electrons-;for example, 2p+, 2n0, 2e-for helium. Which of the following represents the 18O isotope of oxygen?
(A) 7p+,2n0, 9e-
(B) 8p+,10n0, 8e-
(C) 9p+,9n0, 9e-
(D) 10p+,8n0, 9e-
The researchers found that the Neanderthal fossil had approximately 0.0078 as much 14C as found originally in the atmosphere. (a) Using the numbers on your graph, determine how many half-lives have passed since the Neanderthal died. (b) Using your 14C calibration on the x-axis, what is the approximate age of the Neanderthal fossil in years (round off to the nearest thousand)? (c) Approximately when did Neanderthals become extinct according to this study? (d) The researchers cite evidence that modern humans (H. sapiens) became established in the same region as the last Neanderthals approximately 39,000 – 42,000 years ago. What does this suggest about the overlap of Neanderthals and modern humans?
Which coefficients must be placed in the following blanks so that all atoms are accounted for in the products?
\({C_6}{H_{12}}{O_6} \to \_\_\_{C_2}{H_6}{O_{}} + \_\_\_C{O_2}\)
(A)2,1
(B) 3,1
(C) 1,3
(D) 2,2
What do you think about this solution?
We value your feedback to improve our textbook solutions.