Chapter 2: Q2.3-1CC. (page 40)
Why does the structure H-C=C-H fail to make sense chemically?
Short Answer
The H-C=C-H does not make sense chemically because each carbon atom has three covalent bonds in this structure instead of the required four.
Learning Materials
Features
Discover
Chapter 2: Q2.3-1CC. (page 40)
Why does the structure H-C=C-H fail to make sense chemically?
The H-C=C-H does not make sense chemically because each carbon atom has three covalent bonds in this structure instead of the required four.
All the tools & learning materials you need for study success - in one app.
Get started for free
Compared with 31P, the radioactive isotope 32P has
(A) a different atomic number.
(B) one more proton.
(C) one more electron.
(D) one more neutron.
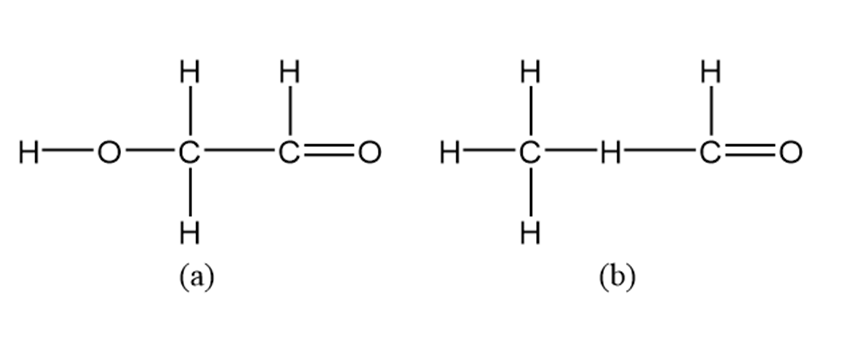
Draw Lewis dot structures for each hypothetical molecule shown below, using the correct number of valence electrons for each atom. Determine which molecules make sense because each atom has a complete valence shell, and each bond has the correct number of electrons. Explain what makes the other molecule nonsensical, considering the number of bonds each type of atom can make.
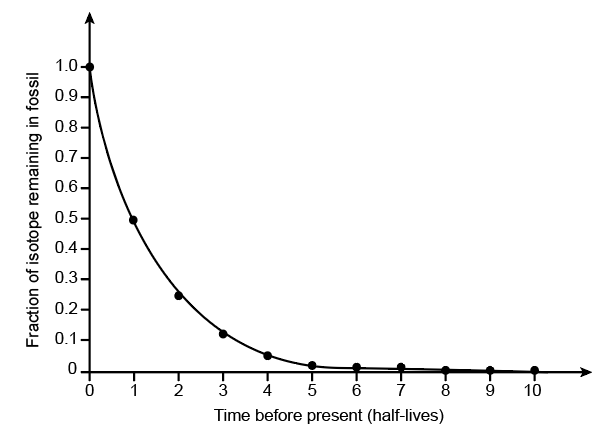
A standard curve of radioactive isotope decay is shown at the top of the right column. The graph line shows the fraction of the radioactive isotope over time (before present) in units of half lives. Recall that a half-life is the amount of time it takes for half of the radioactive isotope to decay. Labeling each data point with the corresponding fractions will help orient you to this graph. Draw an arrow to the data point for half-life = 1 and write the fraction of 14C that will remain after one half life. Calculate the fraction of 14C remaining at each half-life and write the fractions on the graph near arrows pointing to the data points. Convert each fraction to a decimal number and round off to a maximum of three significant digits (zero at the beginning of the number do not count as significant digits). Also write each decimal number in scientific notation.
Is a trace element an essential element? Explain.
A lithium atom has 3 protons and 4 neutrons. What is its mass number?
What do you think about this solution?
We value your feedback to improve our textbook solutions.