Chapter 3: Q6DQ (page 573)
Why is it sometimes possible to loosen caps on screw-top bottles by dipping the capped bottle briefly into hot water?
Short Answer
Due to the difference in volume expansion, the caps get to loosen up.
Learning Materials
Features
Discover
Chapter 3: Q6DQ (page 573)
Why is it sometimes possible to loosen caps on screw-top bottles by dipping the capped bottle briefly into hot water?
Due to the difference in volume expansion, the caps get to loosen up.
All the tools & learning materials you need for study success - in one app.
Get started for free
Real heat engines, like the gasoline engine in a car, always have some friction between their moving parts, although lubricants keep the friction to a minimum. Would a heat engine with completely frictionless parts be 100%efficient? Why or why not? Does the answer depend on whether or not the engine runs on the Carnot cycle? Again, why or why not?
In which situation must you do more work: inflating a balloon at sea level or inflating the same balloon to the same volume at the summit of Mt. McKinley? Explain in terms of pressure and volume change.
When a wet cloth is hung up in a hot wind in the desert, it is cooled by evaporation to a temperature that may beor so below that of the air. Discuss this process in light of the second law of thermodynamics.
The inside of an oven is at a temperature of 200° C (392° F). You can put your hand in the oven without injury as long as you don’t touch anything. But since the air inside the oven is also at 200° C, why isn’t your hand burned just the same?
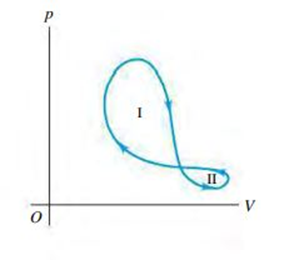
A thermodynamic system undergoes a cyclic process as shown in Fig. Q19.24. The cycle consists of two closed loops: I and II. (a) Over one complete cycle, does the system do positive or negative work? (b) In each loop, is the net work done by the system positive or negative? (c) Over one complete cycle, does heat flow into or out of the system? (d) In each loop, does heat flow into or out of the system? Explain
What do you think about this solution?
We value your feedback to improve our textbook solutions.