Chapter 3: Q46E (page 576)
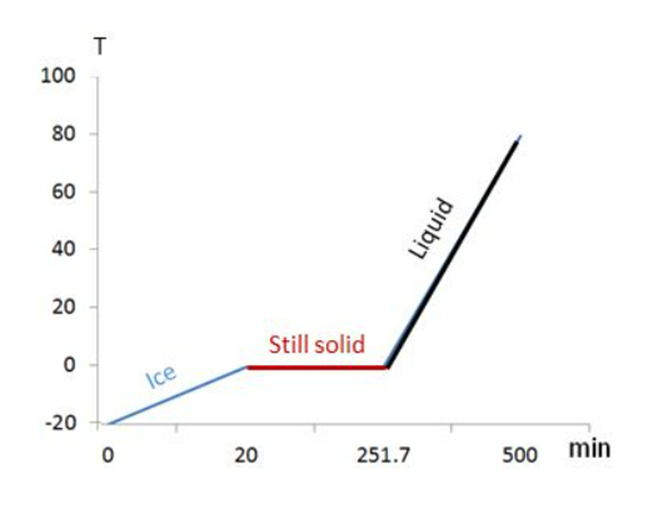
An open container holds 0.550kg of ice at . The mass of the container can be ignored. Heat is supplied to the container at the constant rate 800.0 J/min of for 500.0 min. (a) After how many minutes does the ice start to melt? (b) After how many minutes, from the time when the heating is first started, does the temperature begin to rise above ? (c) Plot a curve showing the temperature as a function of the elapsed time.
Short Answer
(a) t = 21.7 min
(b) t = 251.7 min
(c)