Chapter 3: Q22E (page 611)
A large organic molecule has a mass of . What is the molar mass of this compound?
Short Answer
The Molar Mass of the compound is
Learning Materials
Features
Discover
Chapter 3: Q22E (page 611)
A large organic molecule has a mass of . What is the molar mass of this compound?
The Molar Mass of the compound is
All the tools & learning materials you need for study success - in one app.
Get started for free
Two bodies made of the same material have the same external dimensions and appearance, but one is solid and the other is hollow. When their temperature is increased, is the overall volume expansion the same or different? Why?
On a sunny day, large “bubbles” of air form on the sun-warmed earth, gradually expand, and finally break free to rise through the atmosphere. Soaring birds and glider pilots are fond of using these “thermals” to gain altitude easily. This expansion is essentially an adiabatic process. Why?
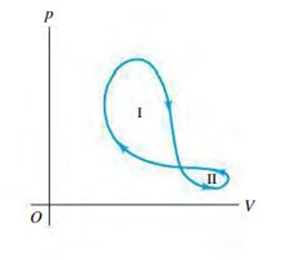
A thermodynamic system undergoes a cyclic process as shown in Fig. Q19.24. The cycle consists of two closed loops: I and II. (a) Over one complete cycle, does the system do positive or negative work? (b) In each loop, is the net work done by the system positive or negative? (c) Over one complete cycle, does heat flow into or out of the system? (d) In each loop, does heat flow into or out of the system? Explain
Compare the pV-diagram for the Otto cycle in Fig. 20.6with the diagram for the Carnot heat engine in Fig. 20.13. Explain some of the important differences between the two cycles.
Section 18.1 states that ordinarily, pressure, volume, and temperature cannot change individually without one affecting the others. Yet when a liquid evaporates, its volume changes, even though its pressure and temperature are constant. Is this inconsistent? Why or why not?
What do you think about this solution?
We value your feedback to improve our textbook solutions.