Chapter 6: Q6DQ (page 1474)
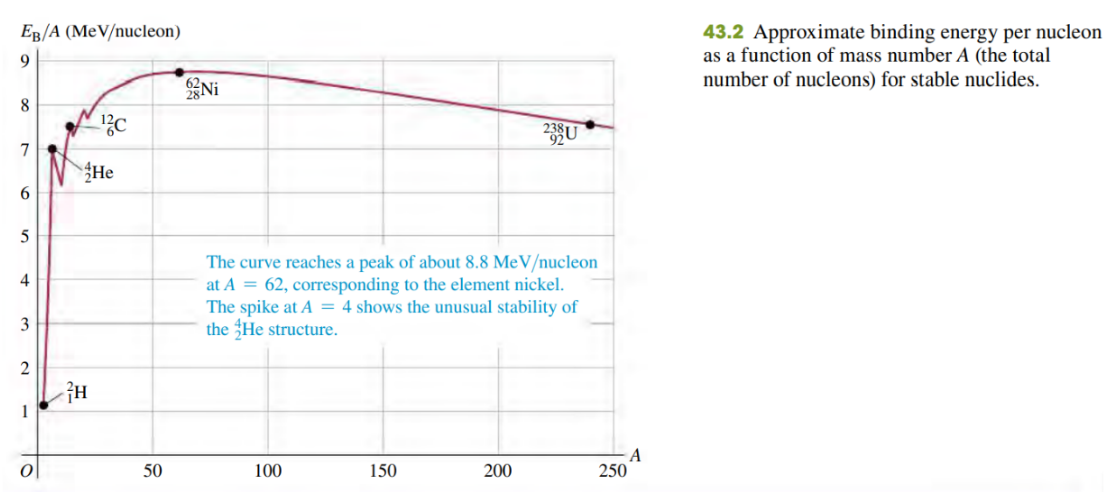
The binding energy per nucleon for most nuclides doesn’t vary much (see Fig. 43.2). Is there similar consistency in the atomic energy of atoms, on an “energy per electron” basis? If so, why? If not, why not?
Short Answer
No, there is no similar consistency in the atomic energy of atoms on an energy per electron basis because the binding energy of electrons is electromagnetic. In contrast, the nucleons are bounded by the strong nuclear force, which does saturate.

