Chapter 6: Q49P (page 1316)
An atom in a metastable state has a lifetime of 5.2 ms. What is the uncertainty in energy of the metastable state?
Short Answer
The minimum uncertainty in energy of the metastable state is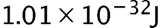
Learning Materials
Features
Discover
Chapter 6: Q49P (page 1316)
An atom in a metastable state has a lifetime of 5.2 ms. What is the uncertainty in energy of the metastable state?
The minimum uncertainty in energy of the metastable state is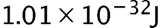
All the tools & learning materials you need for study success - in one app.
Get started for free
In Eq. (43.11), as the total number of nucleons becomes larger, the importance of the second term in the equation decreases relative to that of the first term. Does this make physical sense? Explain.
In what ways do atoms in a diatomic molecule behave as though they were held together by a spring? In what ways is this a poor description of the interaction between the atoms?
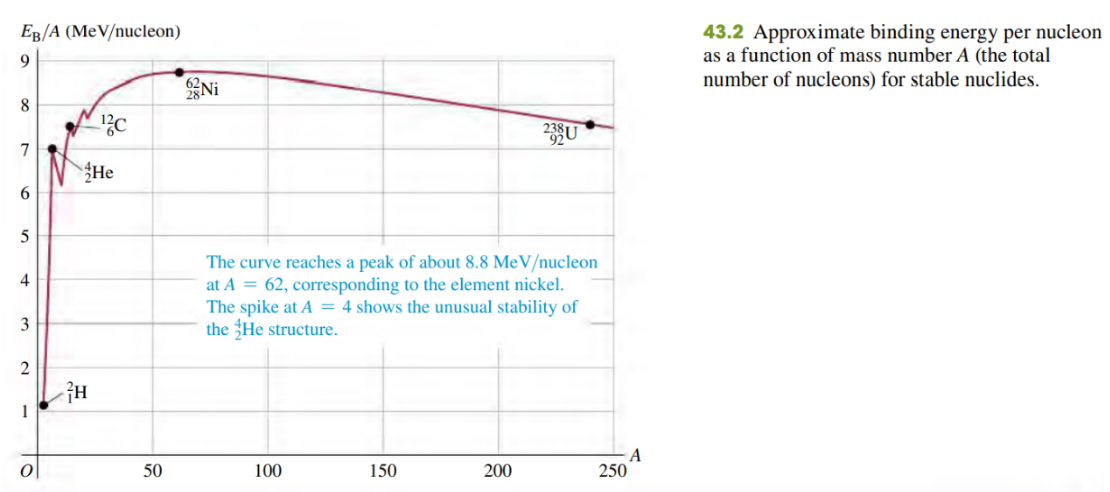
The binding energy per nucleon for most nuclides doesn’t vary much (see Fig. 43.2). Is there similar consistency in the atomic energy of atoms, on an “energy per electron” basis? If so, why? If not, why not?
The wave function shown in Fig. 40.20 is nonzero for both. Does this mean that the particle splits into two parts when it strikes the barrier, with one part tunneling through the barrier and the other part bouncing off the barrier? Explain
According to the standard model of the fundamental particles, what are the similarities between quarks and leptons? What are the most important differences?
What do you think about this solution?
We value your feedback to improve our textbook solutions.